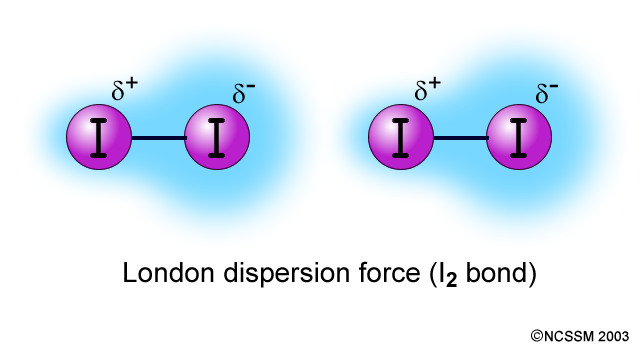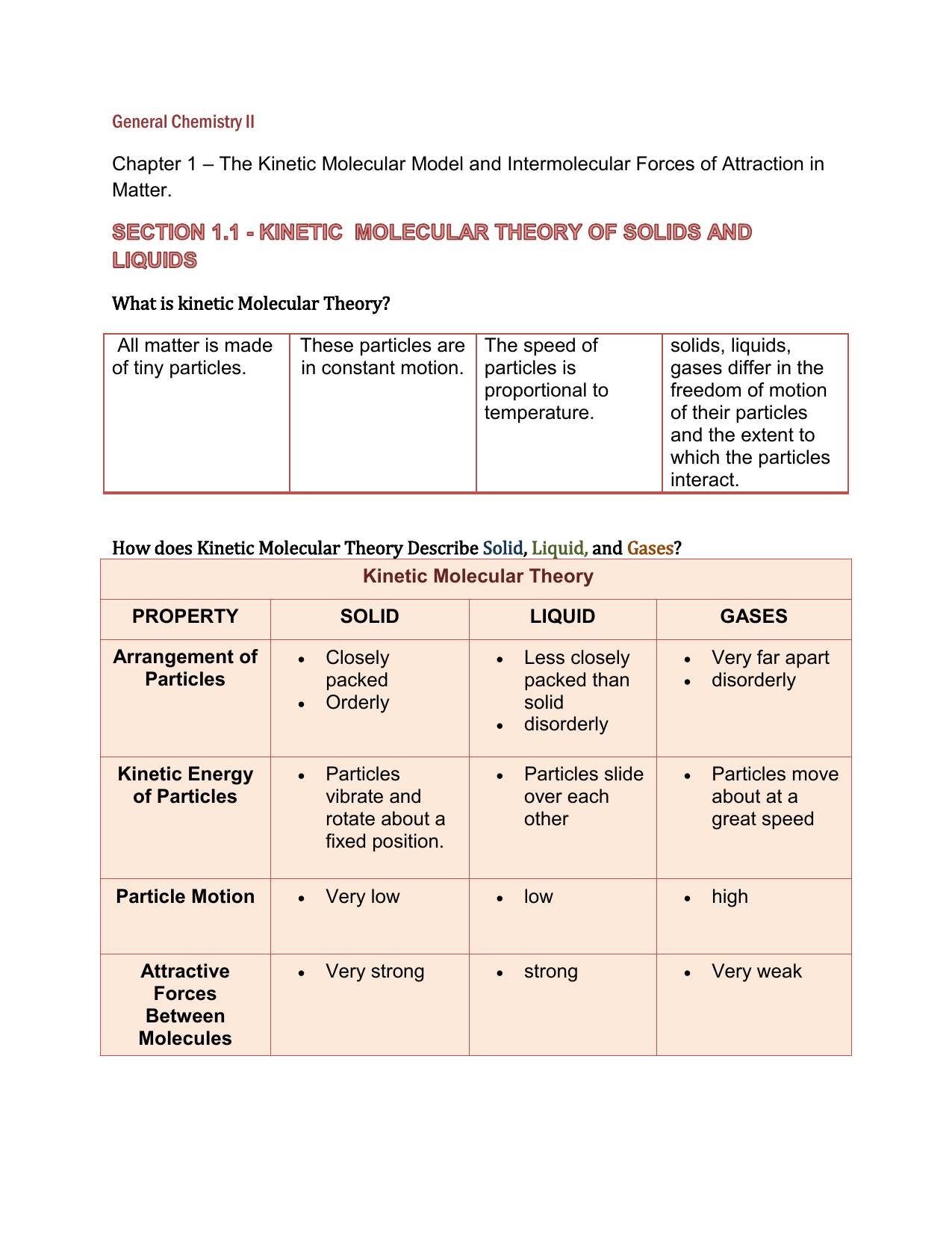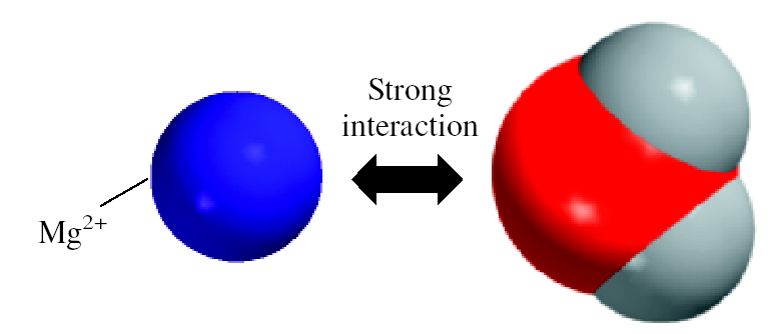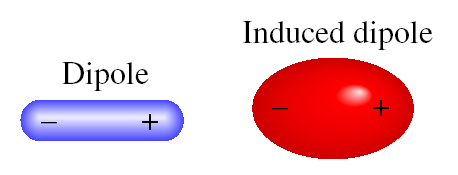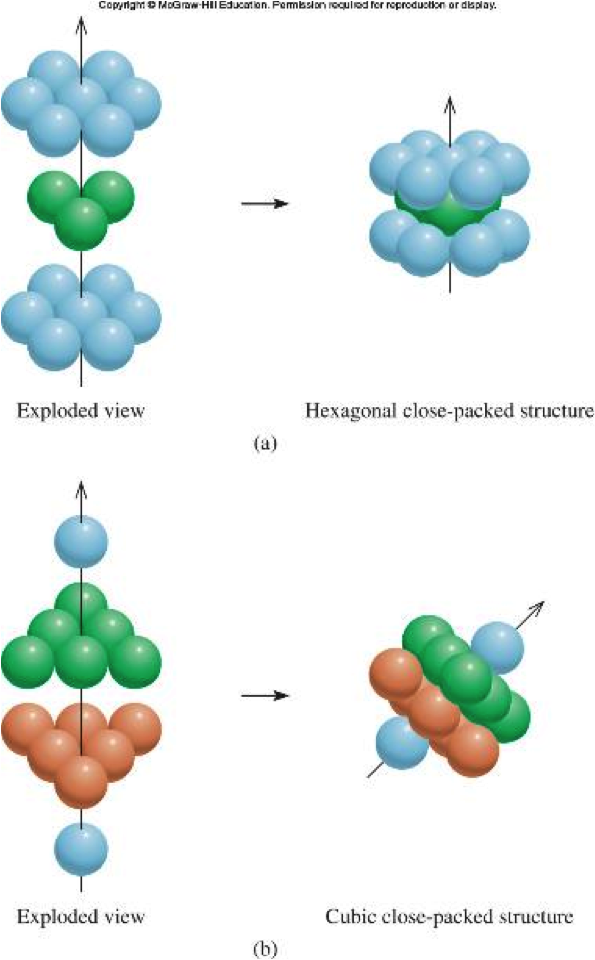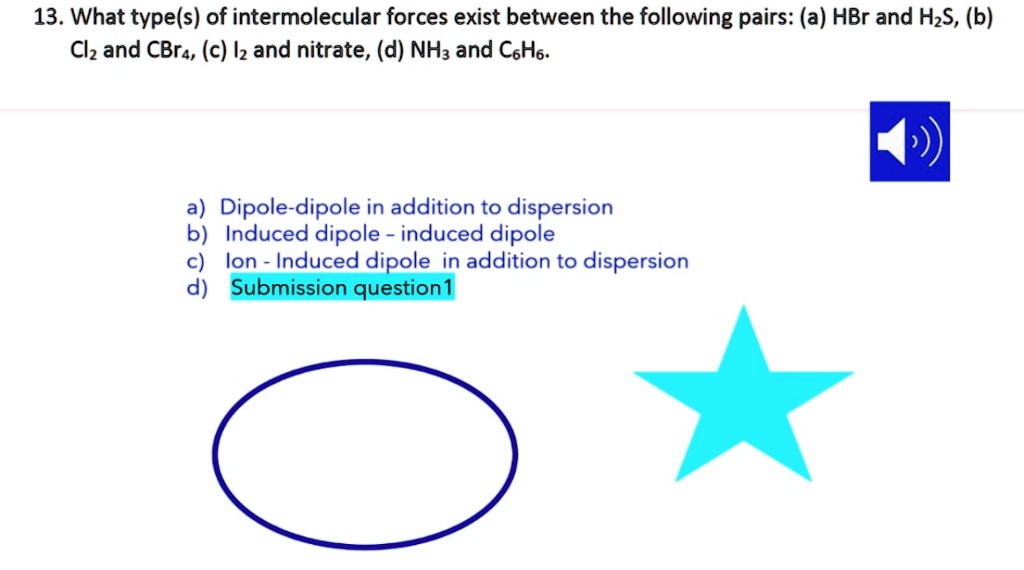
SOLVED: 13. What type(s) of intermolecular forces exist between the following pairs: (a) HBr and HzS, (b) Clz and CBr4, (c) Iz and nitrate; (d) NHz and CsHo: a) Dipole-dipole in addition
![PDF] DFT description of the magnetic structure of polynuclear transition-metal clusters: The complexes [{Cu(bpca)2(H2O)2}{Cu(NO3)2}2], (bpca = Bis(2-pyridylcarbonyl)amine), and [Cu(DBSQ)(C2H5O)]2, (DBSQ = 3,5-di-tert-butyl-semiquinonato) by Alessandro ... PDF] DFT description of the magnetic structure of polynuclear transition-metal clusters: The complexes [{Cu(bpca)2(H2O)2}{Cu(NO3)2}2], (bpca = Bis(2-pyridylcarbonyl)amine), and [Cu(DBSQ)(C2H5O)]2, (DBSQ = 3,5-di-tert-butyl-semiquinonato) by Alessandro ...](https://og.oa.mg/DFT%20description%20of%20the%20magnetic%20structure%20of%20polynuclear%20transition-metal%20clusters%3A%20The%20complexes%20%5B%7BCu(bpca)2(H2O)2%7D%7BCu(NO3)2%7D2%5D%2C%20(bpca%20%3D%20Bis(2-pyridylcarbonyl)amine)%2C%20and%20%5BCu(DBSQ)(C2H5O)%5D2%2C%20(DBSQ%20%3D%203%2C5-di-tert-butyl-semiquinonato).png?author=%20Alessandro%20Bencini,%20Federico%20Totti)
PDF] DFT description of the magnetic structure of polynuclear transition-metal clusters: The complexes [{Cu(bpca)2(H2O)2}{Cu(NO3)2}2], (bpca = Bis(2-pyridylcarbonyl)amine), and [Cu(DBSQ)(C2H5O)]2, (DBSQ = 3,5-di-tert-butyl-semiquinonato) by Alessandro ...

Structural and Bonding Analysis in Monomeric Actinide(IV) Oxalate from Th(IV) to Pu(IV): Comparison with the An(IV) Nitrate Series | Inorganic Chemistry

SOLVED: What type of intermolecular forces exist between the following pairs? (a) HBr and H2S (b) CCl4 and SF6 (c) I2 and NO3- (d) NH3 and H2

Iodine and fluorine form a series of inter halogen molecules and ions. Among these are IF (minute quantities observed spectroscopically), IF_3, IF_4^(-), IF_5, IF_6^(-), and IF_7. (a) Write Lewis formulas for each
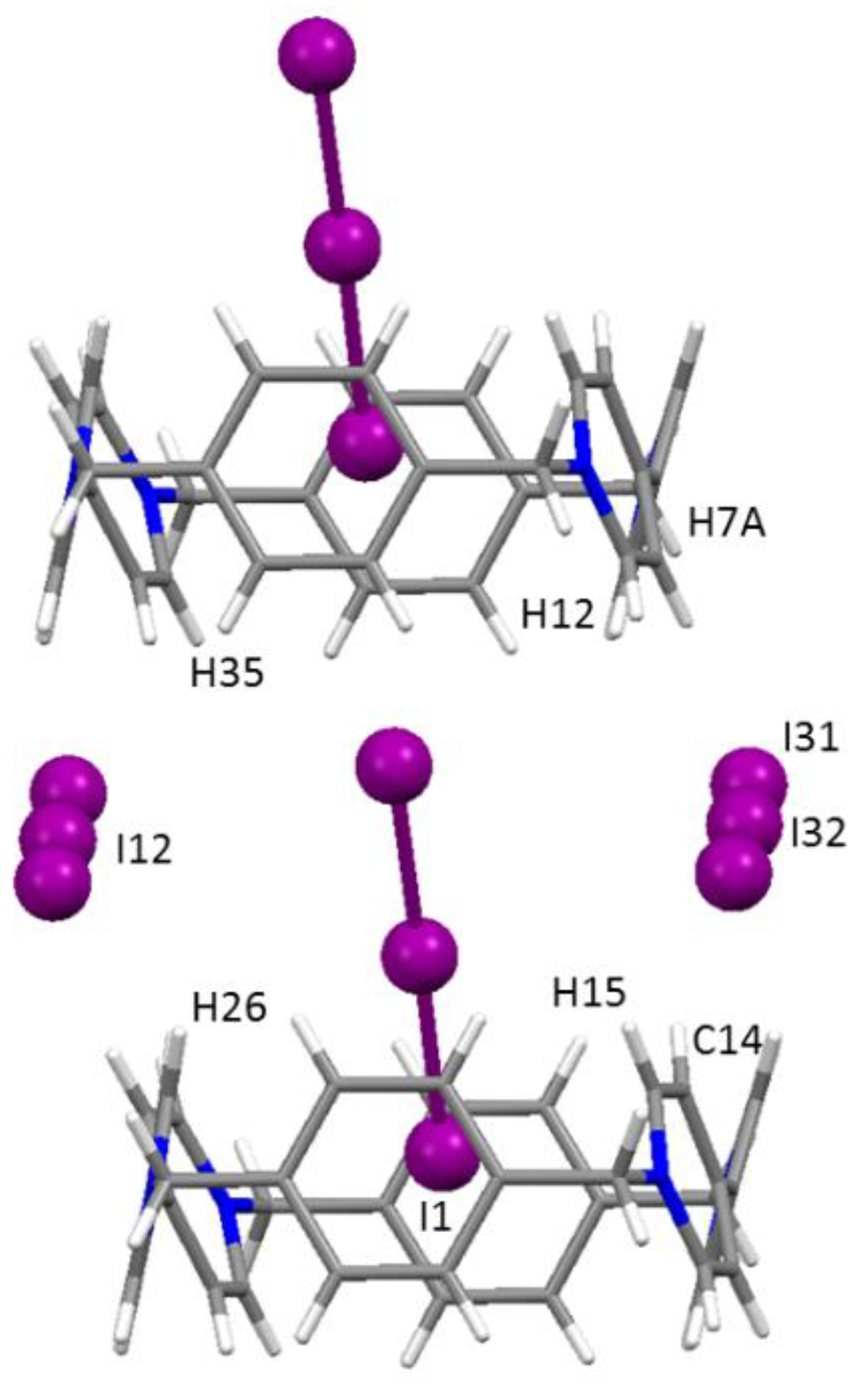
Crystals | Free Full-Text | Genesis of Complex Polyiodide Networks: Insights on the Blue Box/I−/I2 Ternary System

Mediating iodine cathodes with robust directional halogen bond interactions for highly stable rechargeable Zn-I2 batteries - ScienceDirect

a) Complete the following chemical equation : (i) Cu + HNO3 (dil.)→ (ii) XeF4 + O2F2→ (b) Explain the following observations : (i) Phosphorus has a grater tendency for catenation than nitrogen. (

7.28 | Write Lewis structures for the following: H2, HBr, PCl3, SF2, H2CCH2, HNNH, H2CNH, NO-, N2 - YouTube