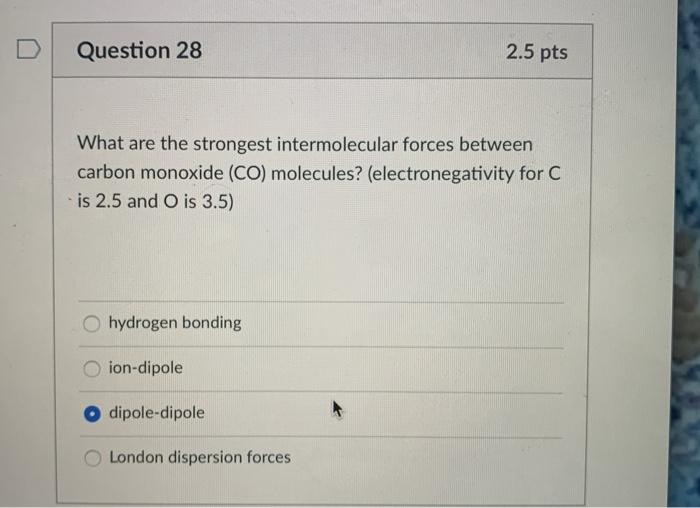
SOLVED: 1. What is the strongest intermolecular force present for each of the following molecules? hydrogen (Hz) carbon monoxide (CO) silicon tetrafluoride (SiF4) D nitrogen tribromide (NBrs) water (HO) acetone (CHzO) methane (

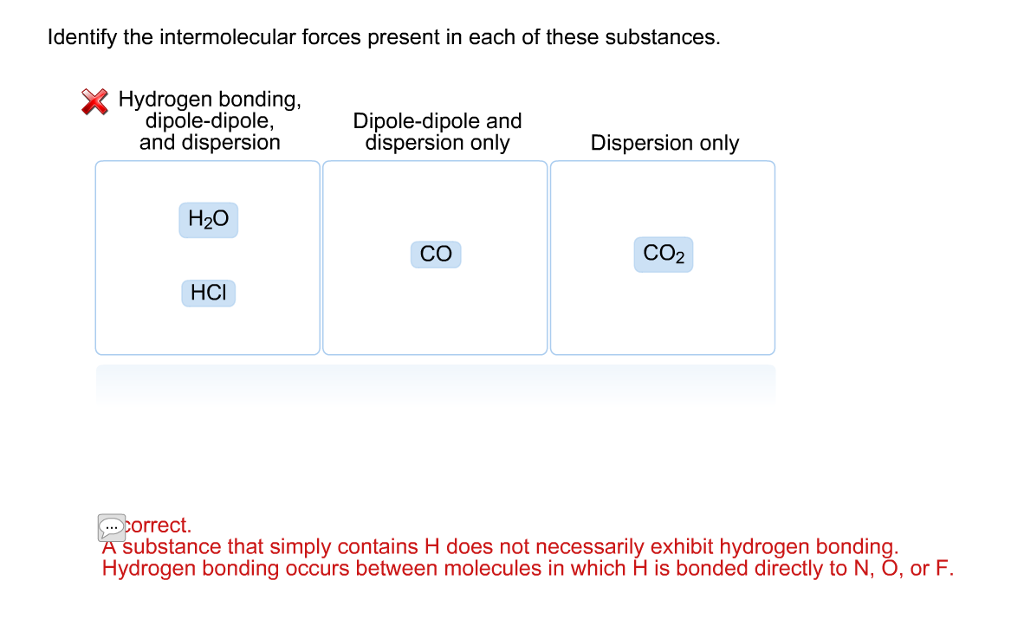
SOLVED: Classify each substance based on the intermolecular forces present in that substance. Hydrogen bonding; dipole-dipole, and dispersion Dipole-dipole and dispersion only Dispersion only HCI I,O CIL; CO Answer Bank Incorrect

Intermolecular-Forces-Worksheet-Key-covalen bonds-and intermolecular forces - Intermolecular Force - Studocu
![12 Chemistry Molecules] Which types of intermolecular forces will occur in this molecule? (Fe(co)5: Iron Pentacarbonyl) : r/HomeworkHelp 12 Chemistry Molecules] Which types of intermolecular forces will occur in this molecule? (Fe(co)5: Iron Pentacarbonyl) : r/HomeworkHelp](https://i.redd.it/xzpgoazopx471.png)
12 Chemistry Molecules] Which types of intermolecular forces will occur in this molecule? (Fe(co)5: Iron Pentacarbonyl) : r/HomeworkHelp
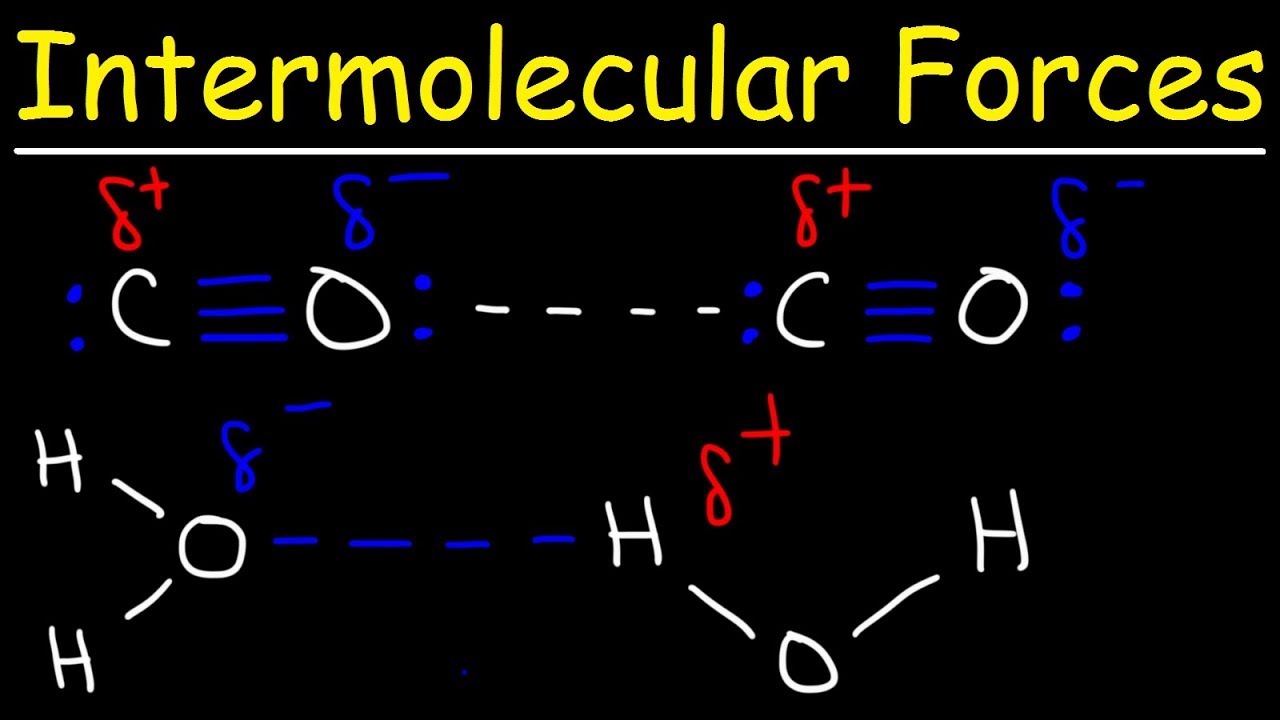
Intermolecular Forces - Hydrogen Bonding, Dipole Dipole Interactions - Boiling Point & Solubility - YouTube
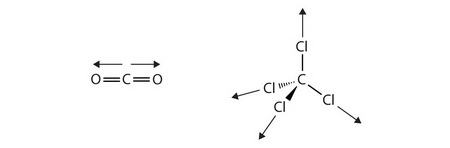
What kind of intermolecular forces are present in the following compounds: C Cl_4, CH_2Cl_2, CH_3OH, CO_2, SCl_4, and SCl_6? | Socratic
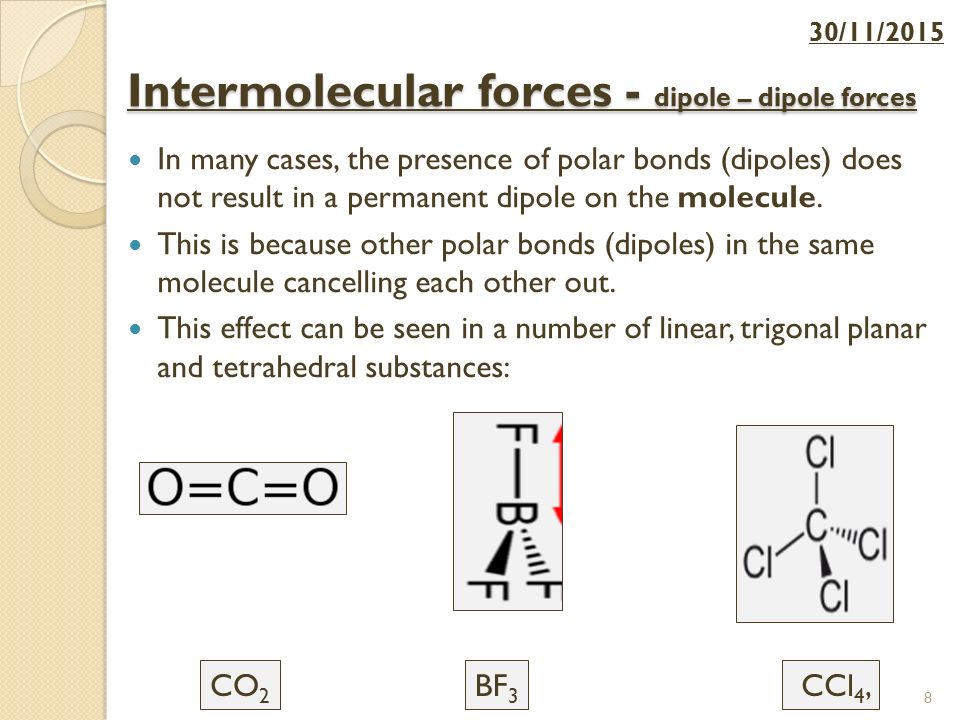
Intermolecular forces – dipole – dipole forces Lesson Objectives: To describe the interaction of molecules by permanent dipole – dipole To compare dipole. - ppt download