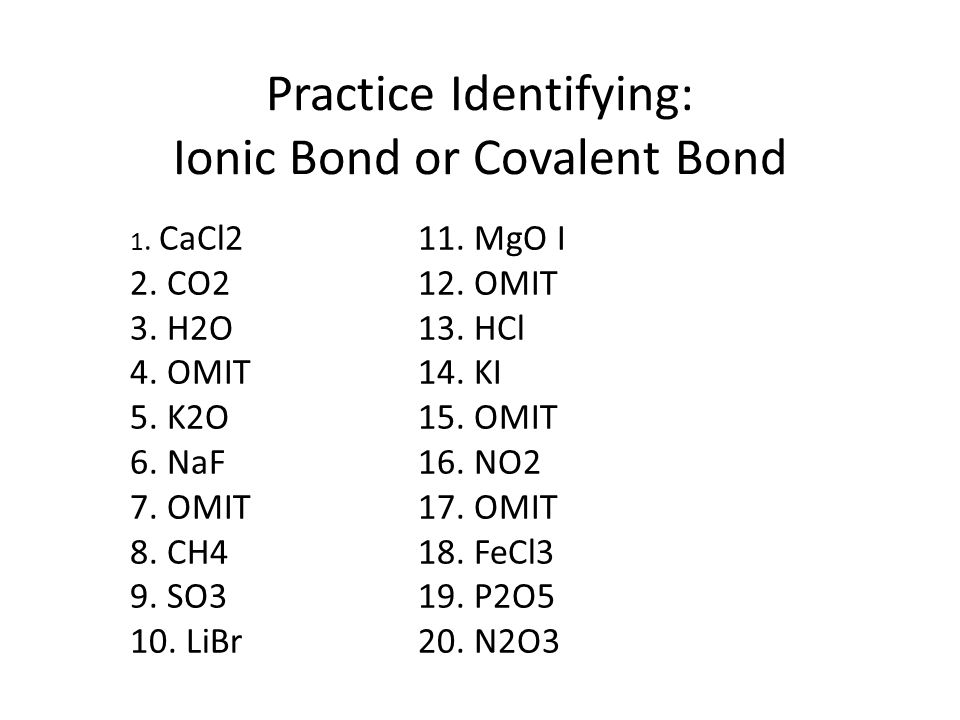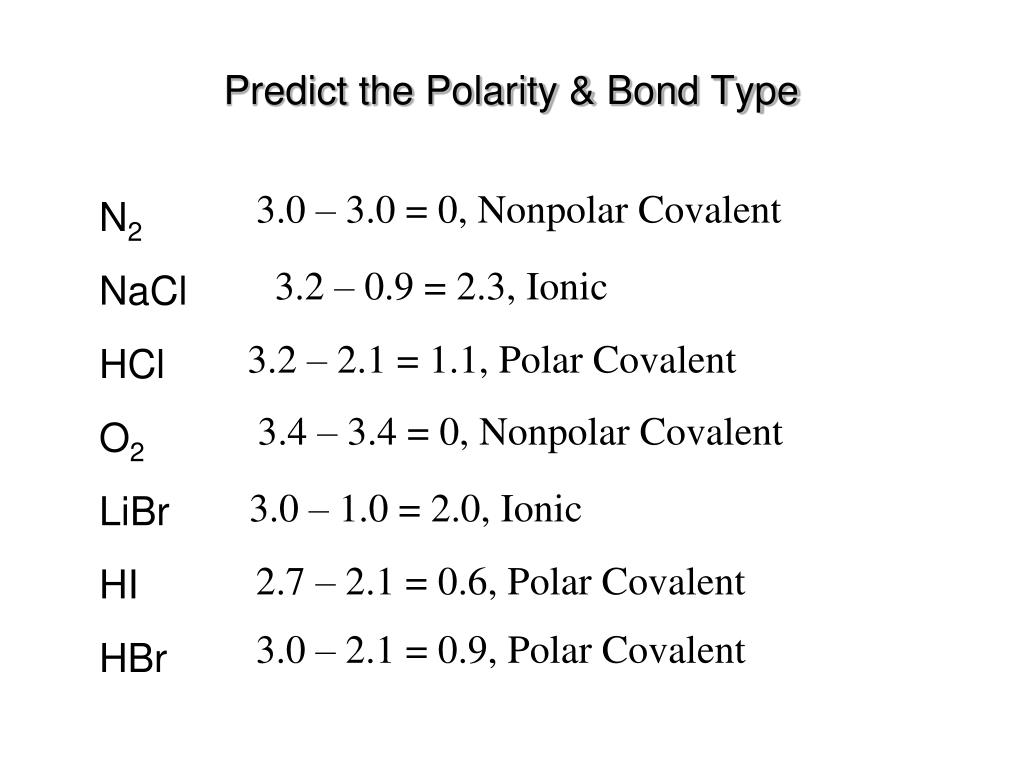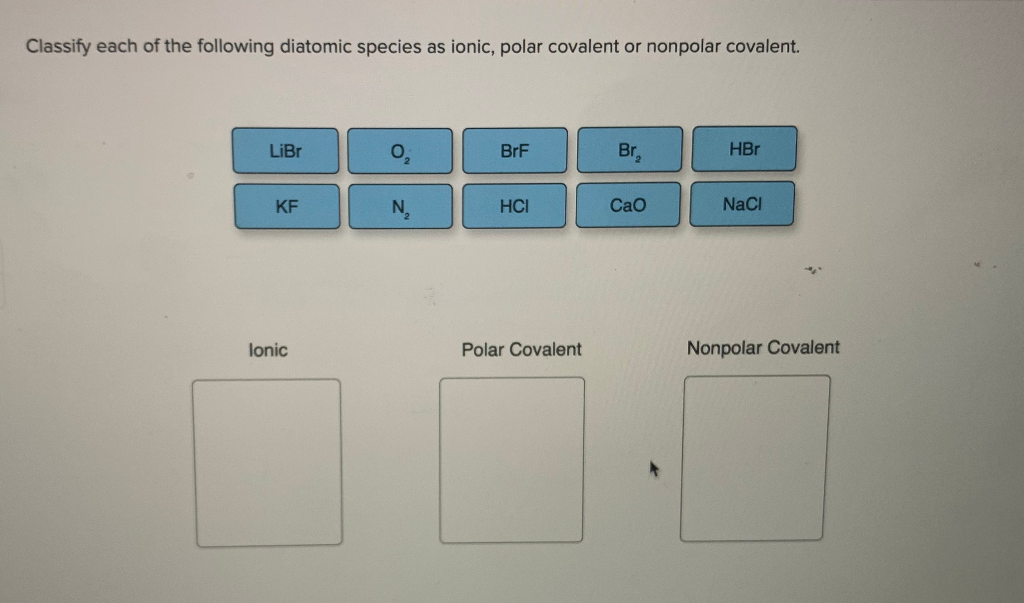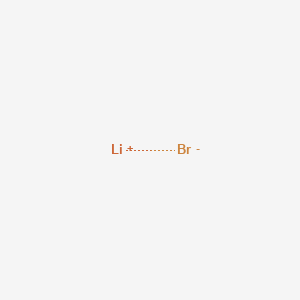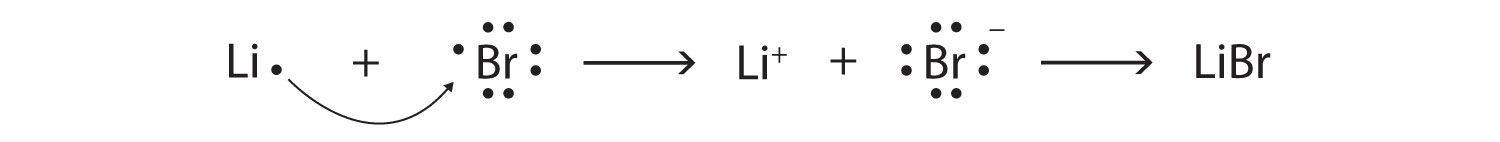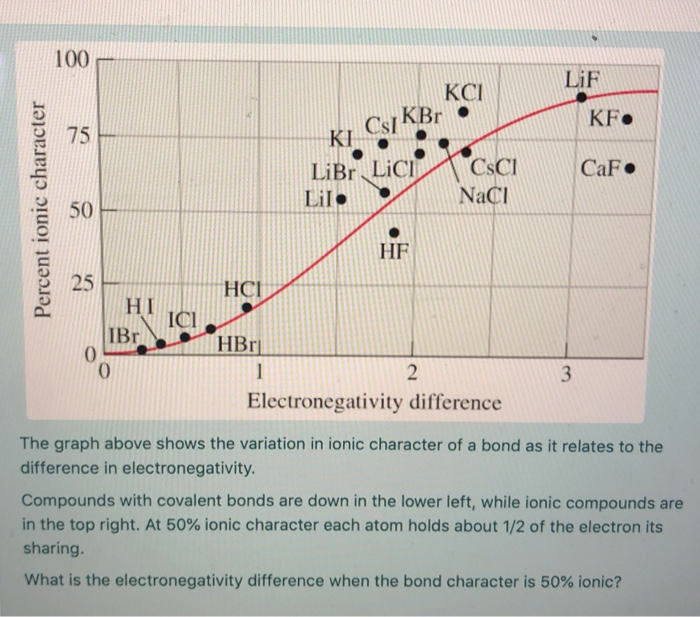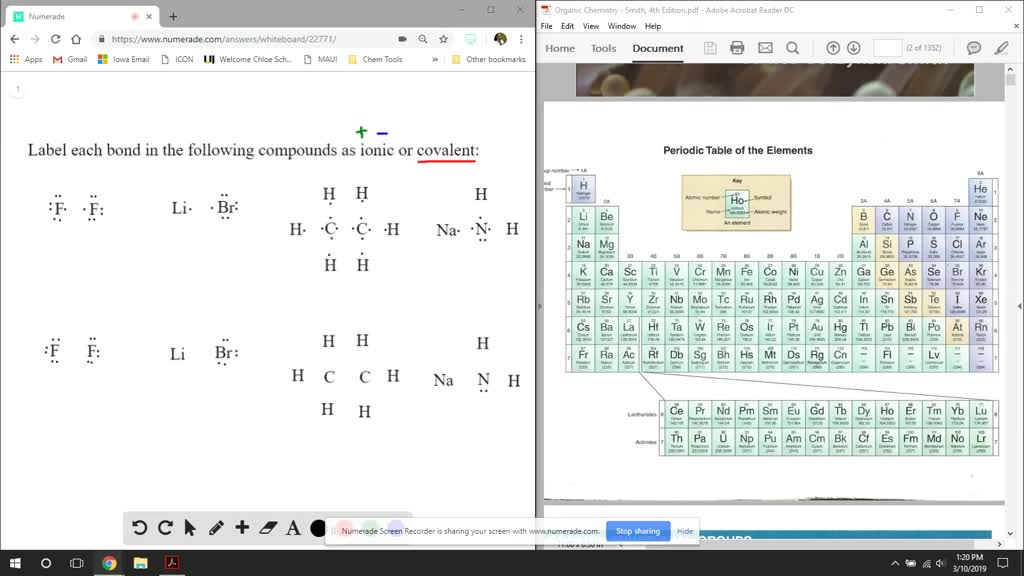
SOLVED:Label each bond in the following compounds as ionic or covalent. a. F2 b. LiBr c. CH3CH3 d. NaNH2
![SOLVED: CHEMIOO Name date L Classifving lonic versus Covalent Lewis Dot Structures of Atoms Classify the following " compounds ionic ([metal covalent (nonmetalt ammonium ion] [non-metal or polyatomic ion]); nonmetal): CaClz COz SOLVED: CHEMIOO Name date L Classifving lonic versus Covalent Lewis Dot Structures of Atoms Classify the following " compounds ionic ([metal covalent (nonmetalt ammonium ion] [non-metal or polyatomic ion]); nonmetal): CaClz COz](https://cdn.numerade.com/ask_images/a107b40b7f8845a78ef0567c586450de.jpg)
SOLVED: CHEMIOO Name date L Classifving lonic versus Covalent Lewis Dot Structures of Atoms Classify the following " compounds ionic ([metal covalent (nonmetalt ammonium ion] [non-metal or polyatomic ion]); nonmetal): CaClz COz

LiCl, LiBr, LiI are covalent as well as ionic why ? | CLASS 10 | CLASSIFICATION OF ELEMENTS AND ... - YouTube

SOLVED:Label each bond in the following compounds as ionic or covalent. a. F2 b. LiBr c. CH3 CH3 d. NaNH 2 e. NaOCH3

Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.(i) Tetra phosphorus decoxide (P4O10) (ii) Ammonium phosphate (NH4)3PO4 (iii) SiC (iv) I2 (v) P4 (vi) Plastic (vii) Graphite (

Unit 6 – Chemical Bonds SPS1b. Compare and contrast ionic bonds in terms of electron position. SPS2b. Predict formulas for stable and binary ionic compounds. - ppt download

LiCl,LiBr,LiI ate covalent as well as ionic why - Chemistry - Classification of Elements and Periodicity in Properties - 13067951 | Meritnation.com

Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.(i) Tetra phosphorus decoxide (P4O10) (ii) Ammonium phosphate (NH4)3PO4 (iii) SiC (iv) I2 (v) P4 (vi) Plastic (vii) Graphite (
Chemical Formula Name Ionic or Covalent AgC2H3O2 Silver acetate Ionic Ba(OH)2 Barium hydroxide Ionic Ca3(PO4)2 Calcium phosphate